Results 1 to 10 of 48
Hybrid View
-
10-29-2013, 09:51 AM #1

I figured it was the steel although i may keep an eye out to see if i can find something like this related to the stones rather then the steel, do you believe some of the stones have a high acidic content?
-
10-30-2013, 01:44 AM #2
-
10-30-2013, 11:48 PM #3

Here's a few facts from my science teacher mate.
Abrading steel increases the chance of oxidation. Add water & this increases further.
The black slurry you see is also rust. Iron oxide can be red or black as we all know from looking at old razors.
The slurry colour may also be due to the composition of different minerals in the stones, possibly acidic . The minerals themselves e.g. Mica, can contain iron to further colour the slurry.
The steel itself is another factor to consider as some steels rust quickly.The white gleam of swords, not the black ink of books, clears doubts and uncertainties and bleak outlooks.
-
10-31-2013, 01:51 AM #4I used Nakayamas for my house



- Join Date
- Aug 2009
- Location
- Des Moines
- Posts
- 8,664
- Blog Entries
- 1
Thanked: 2591
-
10-31-2013, 09:48 AM #5

Just for fun i purchased some PH and Alkalinity test strip, mixed up some slurry and wiped it accross a bunch of stone to see what would happen.
Top mark is Alkalinity (light is lower, dark is higher)
Bottom is PH (light is lower, dark is higher)
1 - Water Only
2 - Belgium Couticle
3 - Tam o Shanter
4 - Random Jnat 1
5 - Random Jnat 2
6 - Random Jnat 3
7 - Random Jnat 4
8 - Random Jnat 5
9 - Vinegar (Acidic results)
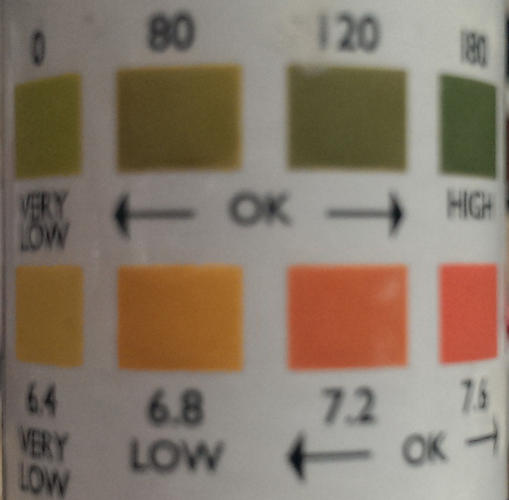

Whats interesting is there is a definite result on some stones although its base, not acidic. Infact, its strong on the base side of the chart.
I have ordered some new strips that have double the range to these, i would love to see how alkaline these are. I will grab some more stones next time as well!Last edited by Brighty83; 10-31-2013 at 10:07 AM. Reason: spelling
-
-
10-31-2013, 10:13 AM #6< Banned User >

- Join Date
- Dec 2012
- Location
- Long Island NY
- Posts
- 1,378
Thanked: 177
Im sure this stone swarf and slurry acidic reaction takes place, but anything that strong would be at least partially neutralized by the water which cannot be too acidic as that would be harmful to the plumbing and I mean the water companies mains which costs big money. I don't leave swarf on my stones and always wipe off and Typically if I leave something for the next day I will do the same.
-
10-31-2013, 04:01 PM #7

A pH rhyme from D. Kolb (1979). Journal of Chemical Education, 56, 53:
For coffee it's 5, for tomatoes it's 4;
While household ammonia is 11 or more.
It's 7 for water, if in a pure state,
But rain water is 6, and sea water is 8.
It's basic at 10, quite acidic at 2,
And well above 7 when litmus turns blue.
Some find it a puzzlement. Doubtless their fog
Has something to do with that negative log.Hur Svenska stålet biter kom låt oss pröfva på.
-
10-31-2013, 11:38 PM #8

Interesting results Brighty. Do any of these random stones cause the rusty slurry syndrome when honing ?
The white gleam of swords, not the black ink of books, clears doubts and uncertainties and bleak outlooks.
-
11-01-2013, 12:45 AM #9

To be honest I have never really looked for it when honing, even if I did see it I wouldn't have put 2 and 2 together lol. I will try to keep any eye out though. Do you have a stone that causes this and can you post a pic of the results?
FYI, the stone that caused the biggest result is this one.

Yaginoshima suita
-
11-01-2013, 04:31 AM #10

Yep. My Yaginoshima Asagi causes rusty slurry. Do you use that Suita on knives ? Fast stone ?
The white gleam of swords, not the black ink of books, clears doubts and uncertainties and bleak outlooks.


 16Likes
16Likes LinkBack URL
LinkBack URL About LinkBacks
About LinkBacks




 Reply With Quote
Reply With Quote

